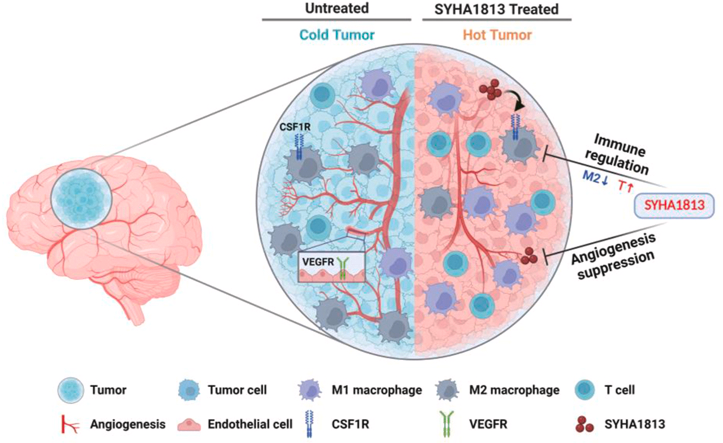
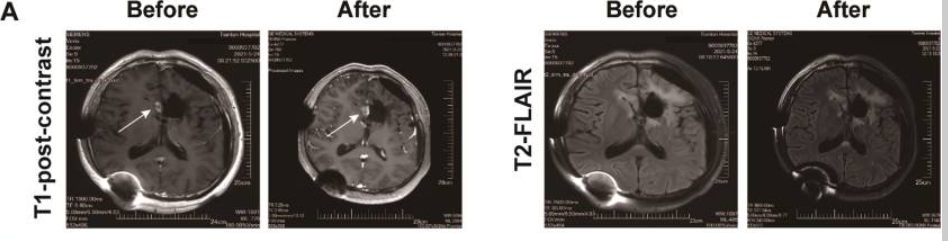
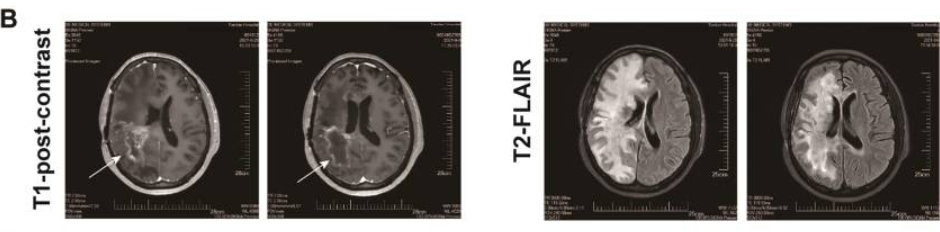
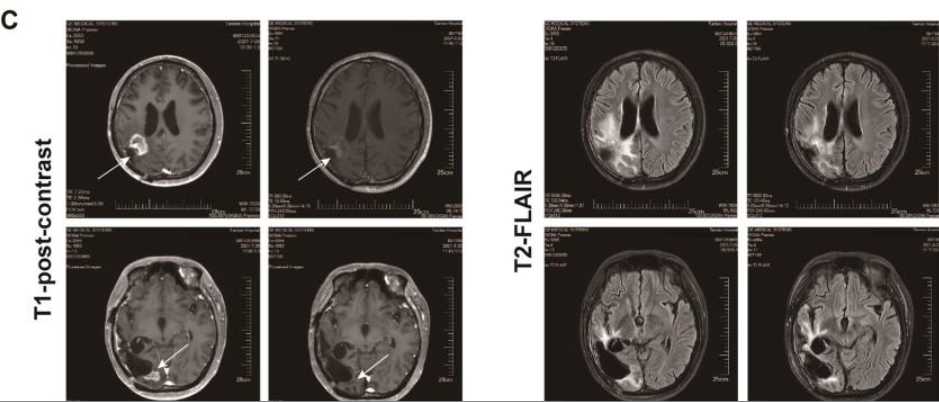
相关推荐
溶瘤病毒是“万能神药”吗?肝癌、膀胱癌、脑瘤都有效?这个“以毒攻毒”的概念为晚期患者按下“暂停键”
2月11日,国际期刊《Journal for ImmunoTherapy of Cancer》发表了溶瘤病毒(ASP9801)用于晚期或转移性实体瘤患者的1期研究结果。
免疫管家 2026-02-13
您的肿瘤,或许可以定制“疫苗”了!肺癌、肝癌、前列腺癌…多种癌症疫苗已取得显著进展
当一名13岁的男孩被诊断为晚期脑胶质瘤并被告知只剩约12个月生命时,他的父亲选择将绝望转化为行动。2015年,Brad Sorenson创立了Providence治疗公司,决心为儿子研发个性化癌症疫苗。如今,他的儿子成功进入了缓解期。
免疫管家 2026-02-12
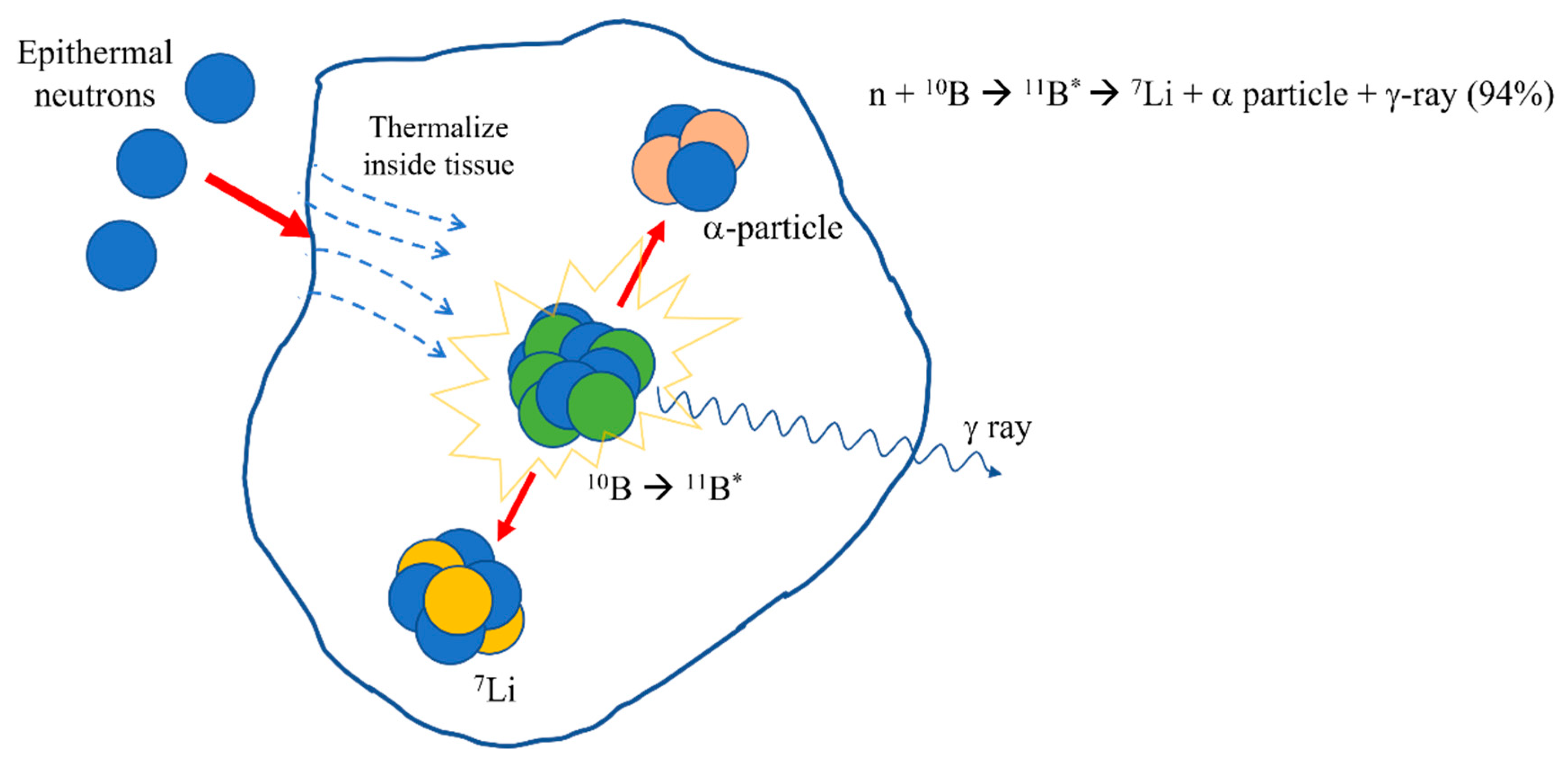
客观缓解率达80.5%!中国硼中子俘获疗法(BNCT)加速落地,国产硼药获突破性认定
2026年1月16日,国家药品监督管理局药品审评中心(CDE)公示了一条重磅消息:中硼(厦门)生物申请的注射用硼[10B]法仑拟纳入突破性治疗,用于既往治疗失败的晚期头颈部恶性肿瘤(不包含鼻咽癌和颅内肿瘤)。
免疫管家 2026-01-20
肿瘤电场治疗是骗局吗?从科学原理到临床数据,一篇说清这项抗癌“黑科技”
近期,我们了解到不少读者朋友有关于肿瘤电场治疗(TTFields)“这是不是骗局?”这样的疑问。面对这种听起来有些“科幻”的新型疗法,产生疑虑是完全可以理解的。今天,我们就来深入浅出地聊一聊肿瘤电场治疗,揭开它的神秘面纱,帮助大家理性看待这项技术。
免疫管家 2026-01-06

Zotiraciclib获得FDA快速通道认定
11月15日,科赛睿生物宣布,美国食品和药物管理局(FDA)已授予Zotiraciclib (ZTR/TG02)快速通道认定,用于治疗携带IDH1或IDH2突变(IDHmut rHGG)的复发高级别脑胶质瘤(rHGG)患者。
免疫管家 2025-11-24

溶瘤病毒Ad-TD-nsIL12为弥漫性脑桥脑胶质瘤(DIPG)患儿带来新希望
7月28日,研究人员在《Nature Communications》发表了溶瘤腺病毒Ad-TD-nsIL12在原发性或进展性小儿IDH野生型弥漫性脑桥脑胶质瘤中的两项I期临床试验结果。
免疫管家 2025-09-25

以毒攻癌的奇迹正在发生!溶瘤病毒为晚期癌症患者开辟一条新的生路,12cm肿瘤近乎消失
长期以来,病毒一直被认为是“敌人”,它们会引发流感、肝炎、艾滋病等疾病,甚至在某些癌症(如人乳头瘤病毒和宫颈癌、EB病毒和鼻咽癌)的发病机制中起到作用。然而,渐渐地,科学家们开始考虑是否有可能驯服病毒,并将这些微小的“杀手”变成攻击癌细胞而不伤害健康细胞的特效剂。这是溶瘤病毒疗法的概念:使用病毒来对抗癌症,并使其准确地...
免疫管家 2025-09-25

癌细胞害怕的"电场疗法"来了!临床实践证实:这种无创治疗让肿瘤生长急刹车
肿瘤电场治疗,顾名思义,通过释放出一种交变电场来干扰癌细胞的活动,让它们没办法正常分裂出新的癌细胞,从而达到抑制肿瘤生长的目的。
免疫管家 2025-04-24

天科雅生物新抗原疫苗治疗弥漫性内生桥脑胶质瘤的Ⅰ期ENACTING研究的初步结果
弥散性内生型脑桥胶质瘤(DIPG)为一种好发于儿童的恶性脑肿瘤,发病后两年内死亡率高于90%。天科雅生物研发了一款新抗原疫苗用于DIPG的治疗,2023年美国临床肿瘤学会(ASCO)年会上公布了新抗原疫苗诱导抗肿瘤T细胞免疫治疗弥漫性内生桥脑胶质瘤的Ⅰ期ENACTING研究的初步结果。
免疫管家 2025-04-09

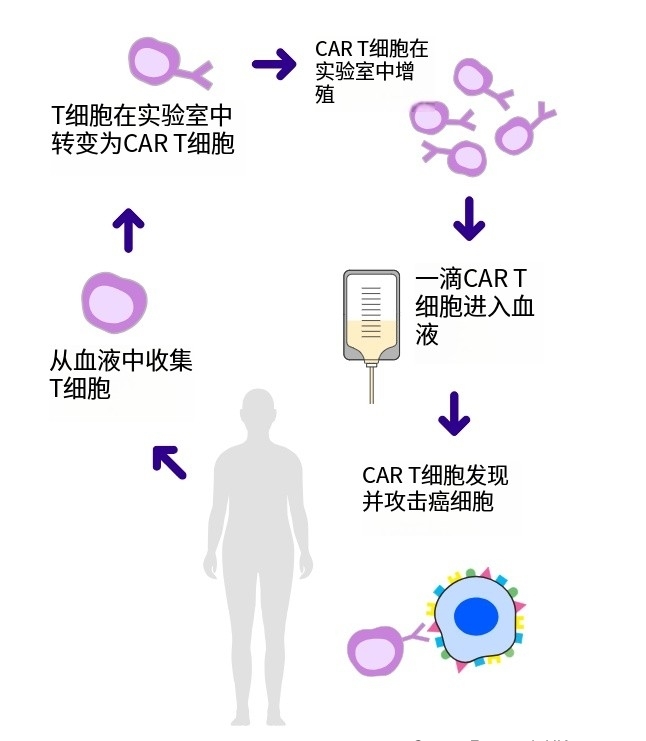
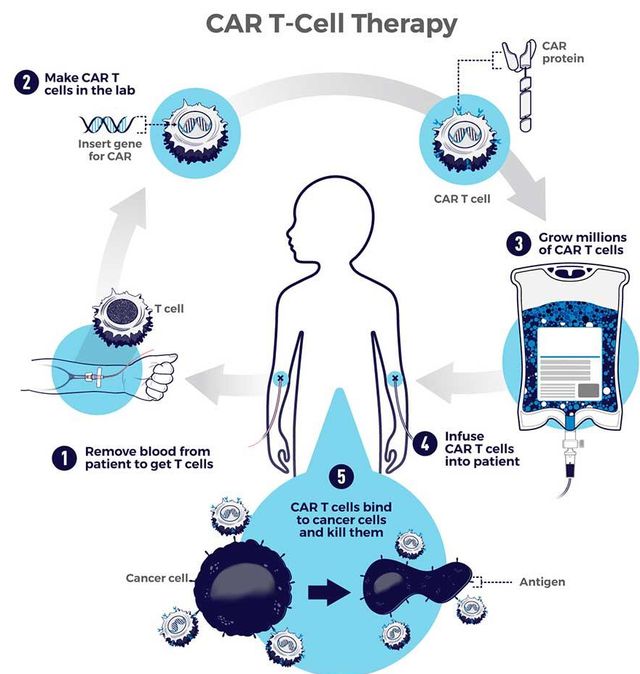
CAR-T细胞疗法在实体瘤领域“大杀四方”,重击胰腺癌、前列腺癌、脑胶质瘤、肾癌
多年来,免疫疗法承载着人类对抗癌症的希望。随着免疫检查点抑制剂、靶向药物、CAR-T细胞疗法等逐渐取得进展,越来越多的癌症患者从中获益,甚至实现长期生存。
免疫管家 2025-04-01